Global Risk Assessment is a user-friendly solution that helps compliance teams schedule, track, and optimize activities.
Digitalize compliance activities. Remediate risks or initiate a formal
investigation. Drive actionable insights with interactive dashboards.
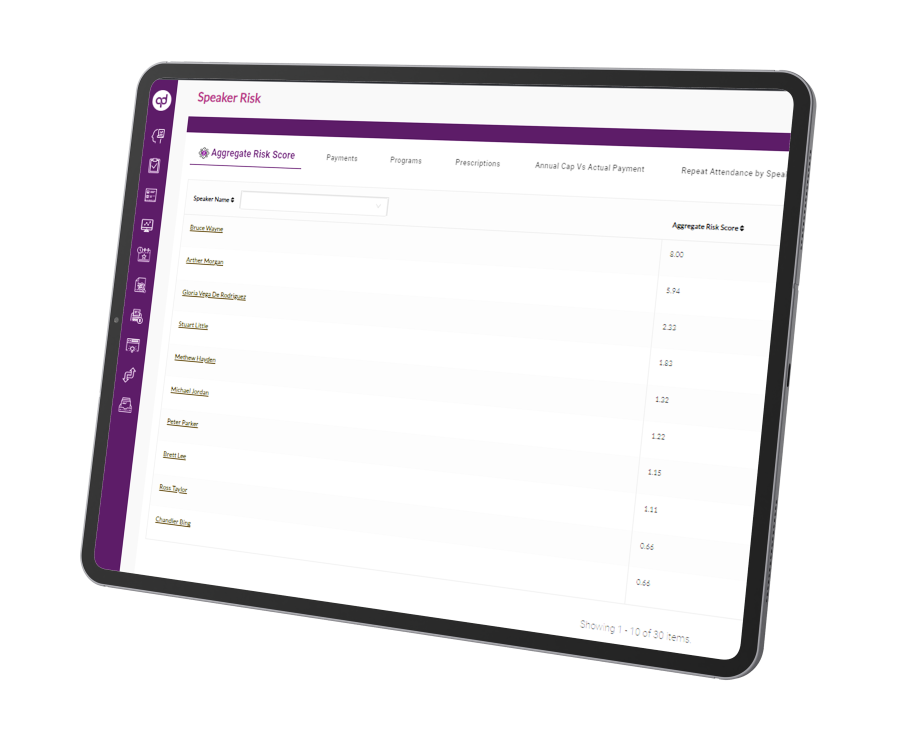
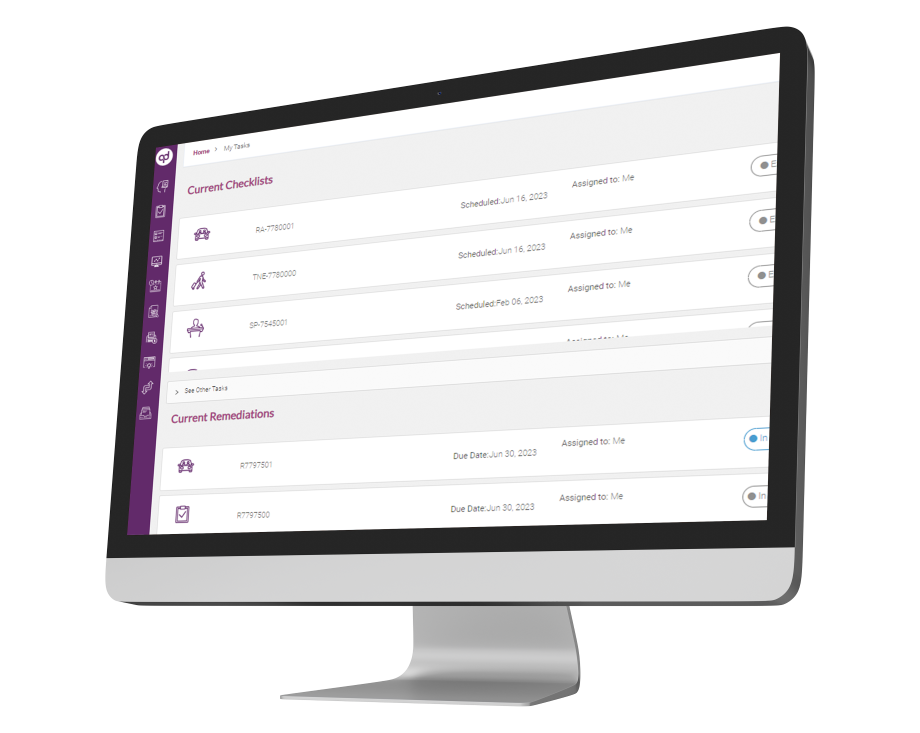
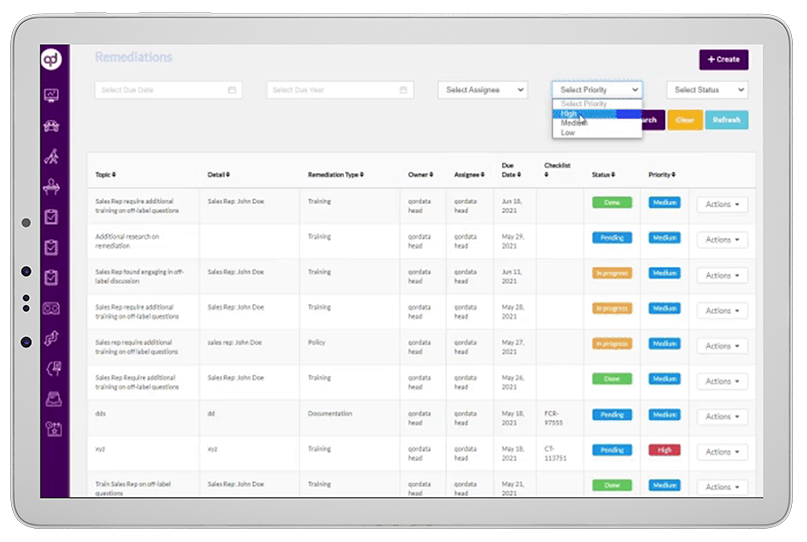
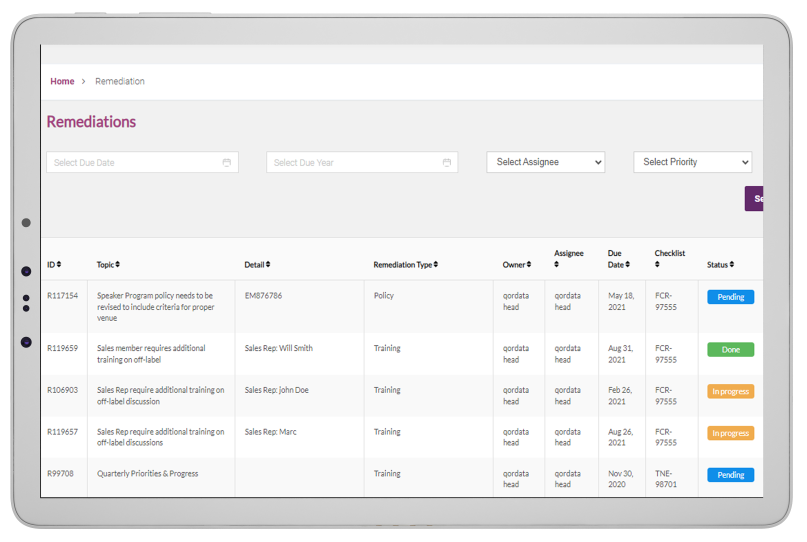
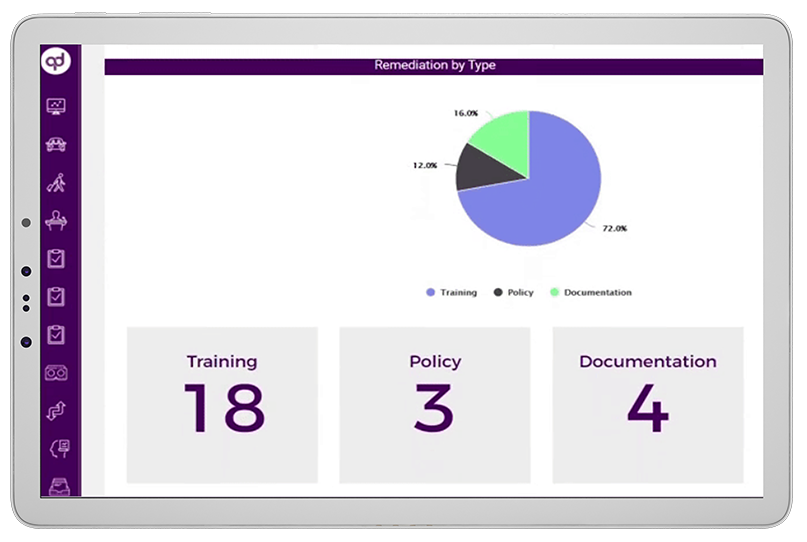
Global Risk Assessment empowers compliance teams to monitor activities throughout the organization and remediate policy violations or business-critical risks from a single user-interface. The solution provides compliance professionals with a digital platform to manage, streamline, monitor and
prioritize compliance issues and take corrective measures to stay compliant.
The U.S Department of Health and Human Services (HHS) Office of Inspector General (OIG) issued the General Compliance Program Guidance (GCPG) ...
Newly appointed compliance professionals cannot be expected to understand the hassle of manually auditing 100% of commercial expense data. With countless ...
“Total penalties from compliance-related violations have crossed over $20 Billion since 2000.” Ethical and law-compliant interaction between life sciences companies and ...
Regulatory expectations and board scrutiny are increasing, and there’s also a growing pressure on compliance leaders to oversee a wider range ...
“Failure to report complete and accurate CMS Open Payments data on time can result in Penalties and Fines of up to ...
CMS Open Payments reporting stands out as a critical aspect of life sciences compliance that demands meticulous attention from compliance officers. ...
© qordata 2024 – All rights reserved